order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
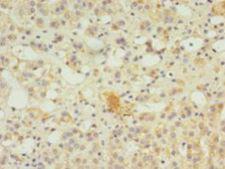
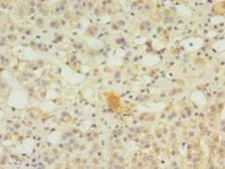
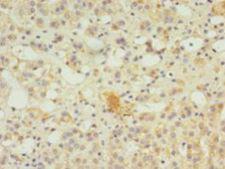
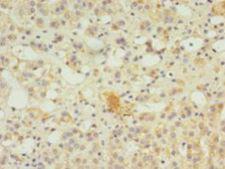
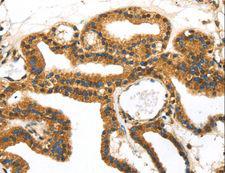
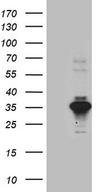
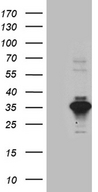
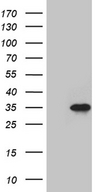
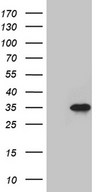
SERPINB8
serpin peptidase inhibitor, clade B (ovalbumin), member 8
The superfamily of high molecular weight serine proteinase inhibitors (serpins) regulate a diverse set of intracellular and extracellular processes such as complement activation, fibrinolysis, coagulation, cellular differentiation, tumor suppression, apoptosis, and cell migration. Serpins are characterized by well-conserved a tertiary structure that consists of 3 beta sheets and 8 or 9 alpha helices (Huber and Carrell, 1989 [PubMed 2690952]). A critical portion of the molecule, the reactive center loop connects beta sheets A and C. Protease inhibitor-8 (PI8; SERPINB8) is a member of the ov-serpin subfamily, which, relative to the archetypal serpin PI1 (MIM 107400), is characterized by a high degree of homology to chicken ovalbumin, lack of N- and C-terminal extensions, absence of a signal peptide, and a serine rather than an asparagine residue at the penultimate position (summary by Bartuski et al., 1997 [PubMed 9268635]).[supplied by OMIM, Jan 2010]
| Gene Name: | serpin peptidase inhibitor, clade B (ovalbumin), member 8 |
| Synonyms: | SERPINB8, Cytoplasmic antiproteinase 2, Peptidase inhibitor 8, PI8, Serpin B8, PI-8, CAP-2 |
| Target Sequences: | NM_002640 NP_002631.3 P50452 |

If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.









