order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
RNGTT / HCAP
RNA guanylyltransferase and 5'-phosphatase
RNGTT is a non-receptor like tyrosine phosphatase. Mammalian capping enzymes are bifunctional proteins with both RNA 5 '- triphosphatase and guanylyltransferase activities. The N-terminal 237- aa triphosphatase domain contains (I/V)HCXXGXXR(S/T)G, a sequence corresponding to the conserved active-site motif in protein tyrosine phosphatases (PTPs). mRNA capping requires the sequential action of three enzymatic activities: RNA triphosphatase, guanylyl-transferase, and methyltransferase. The m7GpppN cap of eukaryotic mRNA is formed by a series of three enzymatic reactions in which the 5'-triphosphate end of nascent pre-mRNA is hydrolyzed to a 5'-diphosphate by RNA triphosphatase, then capped with GMP by GTP: RNA guanylyltransferase, and methylated by RNA (guanine-N7) methyltransferase. The human mRNA capping enzyme has two alternatively spliced isoforms of 597 and 541 aa that differ in their C-terminus.
| Gene Name: | RNA guanylyltransferase and 5'-phosphatase |
| Family/Subfamily: | Protein Phosphatase , Protein Phosphatase - Tyrosine non-receptor like other |
| Synonyms: | RNGTT, CAP1A, HCAP, HCAP1, HCE, HCE1, MRNA-capping enzyme |
| Target Sequences: | NM_003800 NP_003791.3 O60942 |
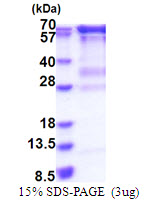
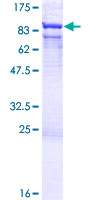
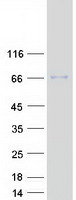
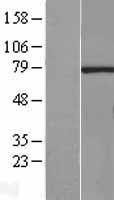
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.









