order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Location
Corporate Headquarters
Vector Laboratories, Inc.
6737 Mowry Ave
Newark, CA 94560
United States
Telephone Numbers
Customer Service: (800) 227-6666 / (650) 697-3600
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
NARF
nuclear prelamin A recognition factor
Several proteins have been found to be prenylated and methylated at their carboxyl-terminal ends. Prenylation was initially believed to be important only for membrane attachment. However, another role for prenylation appears to be its importance in protein-protein interactions. The only nuclear proteins known to be prenylated in mammalian cells are prelamin A- and B-type lamins. Prelamin A is farnesylated and carboxymethylated on the cysteine residue of a carboxyl-terminal CaaX motif. This post-translationally modified cysteine residue is removed from prelamin A when it is endoproteolytically processed into mature lamin A. The protein encoded by this gene binds to the prenylated prelamin A carboxyl-terminal tail domain. It may be a component of a prelamin A endoprotease complex. The encoded protein is located in the nucleus, where it partially colocalizes with the nuclear lamina. It shares limited sequence similarity with iron-only bacterial hydrogenases. Alternatively spliced transcript variants encoding different isoforms have been identified for this gene, including one with a novel exon that is generated by RNA editing.
| Gene Name: | nuclear prelamin A recognition factor |
| Synonyms: | NARF, IOP2 |
| Target Sequences: | NM_012336 NP_036468.1 Q9UHQ1 |
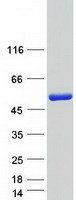
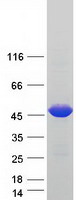
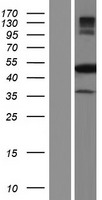
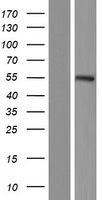
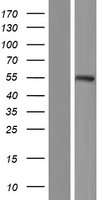
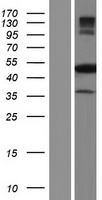
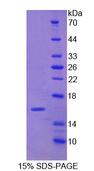
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.










