order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
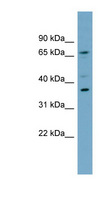
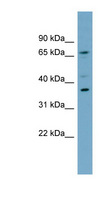
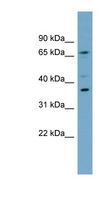
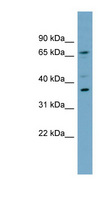
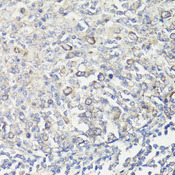
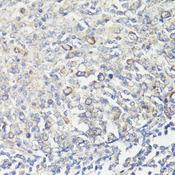
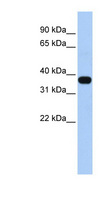
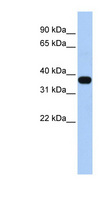
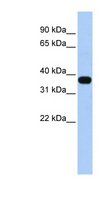
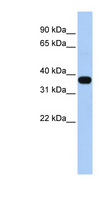
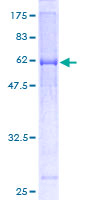
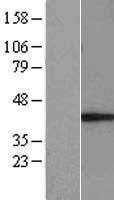
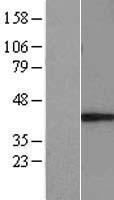
MUL1 / MULAN
mitochondrial E3 ubiquitin protein ligase 1
Exhibits weak E3 ubiquitin-protein ligase activity. E3 ubiquitin ligases accept ubiquitin from an E2 ubiquitin-conjugating enzyme in the form of a thioester and then directly transfer the ubiquitin to targeted substrates. Can ubiquitinate AKT1 preferentially at 'Lys-284' involving 'Lys-48'-linked polyubiquitination and seems to be involved in regulation of Akt signaling by targeting phosphorylated Akt to proteosomal degradation. Proposed to preferentially act as a SUMO E3 ligase at physiological concentrations. Plays a role in the control of mitochondrial morphology. Promotes mitochondrial fragmentation and influences mitochondrial localization. The function may implicate its abilty to sumoylate DNM1L. Inhibits cell growth. When overexpressed, activates JNK through MAP3K7/TAK1 and induces caspase-dependent apoptosis. Involved in the modulation of innate immune defense against viruses by inhibiting DDX58-dependent antiviral response. Can mediate DDX58 sumoylation and disrupt its polyubiquitination.
| Gene Name: | mitochondrial E3 ubiquitin protein ligase 1 |
| Synonyms: | MUL1, C1orf166, E3 SUMO-protein ligase MUL1, E3 ubiquitin ligase, MULAN, RING finger protein 218, MAPL, RNF218, GIDE, RP11-401M16.2 |
| Target Sequences: | NM_024544 NP_078820.2 Q969V5 |




If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.









